
Much work has been done with traditional 3D multicellular spheroid models to demonstrate and improve the relevance of these 3D models over 2D culture 12, 13, 14, to assess the delivery and efficacy of therapeutics 15, 16, and to enable HT-HC screening through standardization and automation 17, 18, 19.
#MODEL A LABEL IN SOLIDWORKS 2005 FULL#
Additionally, to achieve their full potential, in vitro phenotypic models should move beyond single population-level outcome measures, such as cytotoxicity, and incorporate high-content (HC) multiplex analyses of various relevant cellular processes and behaviors 10, 11. automated imaging systems, liquid handling robots) 7, 10. For such models to gain traction and widespread use within both pharmaceutical and academic environments, they must be user-friendly, time-efficient, reproducible within and between laboratories, standardizable, scalable, and ideally, amenable to high-throughput (HT) automation (e.g. Specifically, while there is advocacy that added model complexity (inclusion of vasculature, various stromal and immune cells) may increase pathophysiologic relevance and predictive power, such approaches fall short with respect to practical logistics 7, 8, 9. When developing next-generation in vitro phenotypic models of tumor invasion, a number of design criteria should be considered. Altogether, this points to a need for next-generation preclinical tumor-tissue invasion models that effectively recreate key features of the metastatic phenotype and this desmoplastic microenvironment. For PDAC, in particular, deposition of highly-crosslinked, fibrillar type I collagen by cancer associated fibroblasts (CAFs), also known as desmoplasia, represents a prominent ECM-associated change that has been implicated as a promoter of metastasis and a negative prognostic indicator 6.
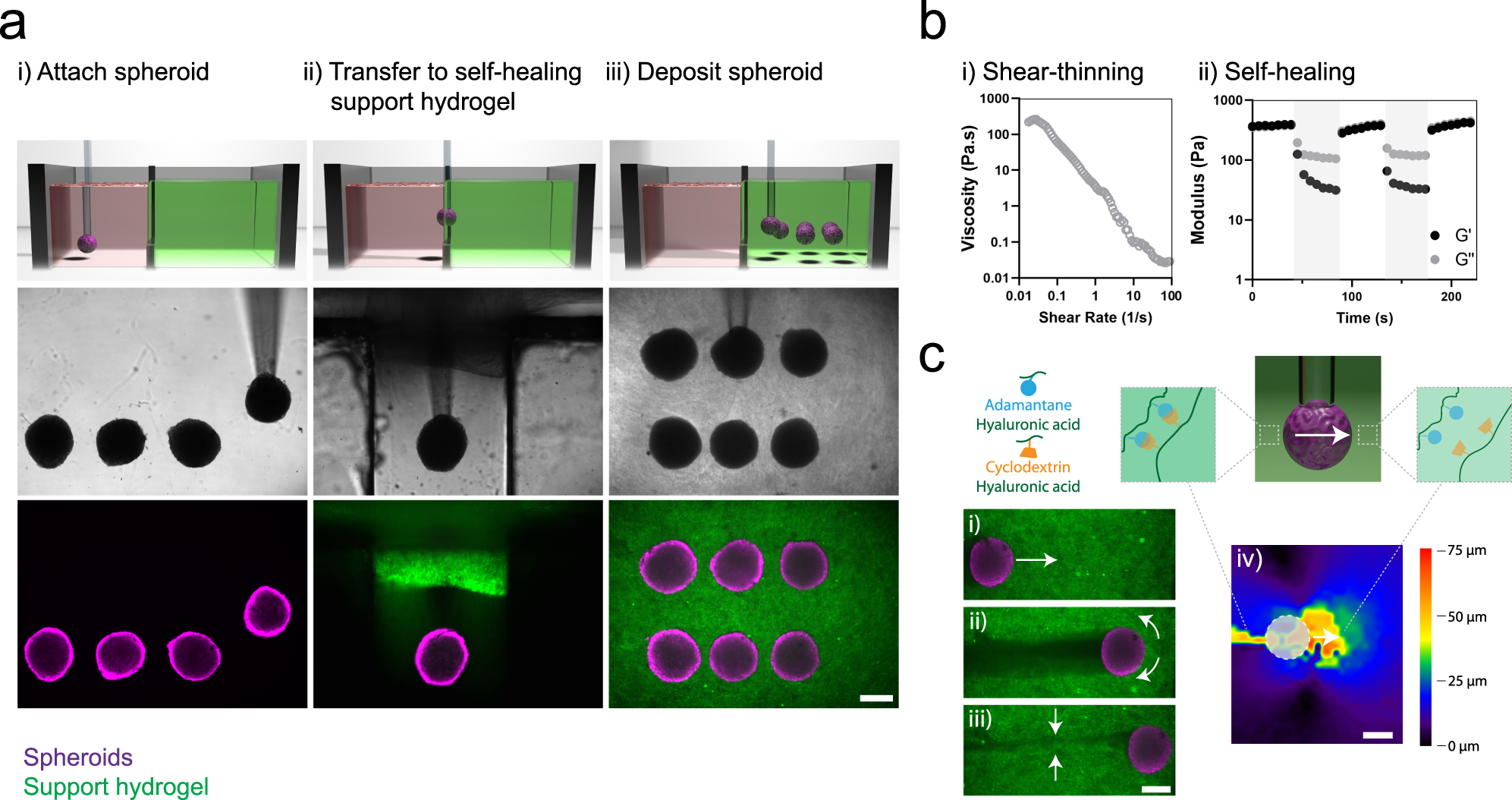
Since the initial step in the tumor metastatic process involves tumor cell engagement, remodeling, and invasion of the surrounding tissue extracellular matrix (ECM), it is becoming increasingly clear that accurate recreation of such three-dimensional (3D) tumor-tissue ECM interactions and associated physicochemical signaling is critical to the development of more pathophysiologically relevant and predictive in vitro models 4, 5. While limited treatment options for patients with metastases represents a multi-facetted problem, one major shortcoming is the lack of predictive preclinical models of invasive tumor phenotypes that can be used for mechanistic studies, biomarker and drug target identification, and drug screening 1, 3.

This problem is especially apparent for highly metastatic cancers like pancreatic ductal adenocarcinoma (PDAC), where approximately 90% of patients present with invasive or metastatic disease 2. Additionally, a proof-of-concept multiplex drug screening assay was applied to highlight this model’s ability to interface with automated imaging systems and showcase its potential as a predictive tool for high-throughput, high-content drug screening.ĭespite progress in treating some cancers, metastatic tumors remain nearly impossible to treat, thus metastasis continues to be the predominant cause of cancer-related deaths 1. Low-passage, patient-derived pancreatic cancer cells and cancer-associated fibroblasts were used to increase model pathophysiologic relevance, yielding fibroblast-mediated tumor invasion and matrix alignment. Pancreatic cancer cell lines used within this model showed expected differential invasive phenotypes. High-cell density tumor compartments were created using a custom-designed fabrication system and standardized oligomeric type I collagen to define and modulate ECM physical properties. To address this gap, a novel 3D tumor-tissue invasion model which supports rapid, reproducible setup and user-definition of tumor and surrounding tissue compartments was developed.
While present-day 3D spheroid invasion models more accurately recreate tumor invasion than traditional 2D models, their shortcomings include poor reproducibility and inability to interface with automated, high-throughput systems. One shortcoming of the drug development process that underlies these problems is the lack of predictive, pathophysiologically relevant preclinical models of invasive tumor phenotypes.
#MODEL A LABEL IN SOLIDWORKS 2005 TRIAL#
While much progress has been made in the war on cancer, highly invasive cancers such as pancreatic cancer remain difficult to treat and anti-cancer clinical trial success rates remain low.
